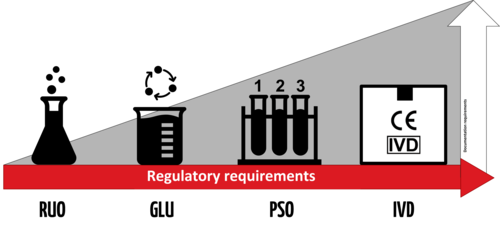
![Laboratory-Developed Tests: Design of a Regulatory Strategy in Compliance with the International State-of-the-Art and the Regulation (EU) 2017/746 (EU IVDR [In Vitro Diagnostic Medical Device Regulation]) | SpringerLink Laboratory-Developed Tests: Design of a Regulatory Strategy in Compliance with the International State-of-the-Art and the Regulation (EU) 2017/746 (EU IVDR [In Vitro Diagnostic Medical Device Regulation]) | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs43441-021-00323-7/MediaObjects/43441_2021_323_Fig2_HTML.png)
Laboratory-Developed Tests: Design of a Regulatory Strategy in Compliance with the International State-of-the-Art and the Regulation (EU) 2017/746 (EU IVDR [In Vitro Diagnostic Medical Device Regulation]) | SpringerLink

Elexes Medical Consulting on Twitter: "RUO - A Market Opportunity for IVD Companies Read more on: https://t.co/bhmwtZ5ZVX #elexes #ivd #regulatory #documentation #labeling #qms #fda #reporting #clinicaldata #manufacturer #performance #usability ...