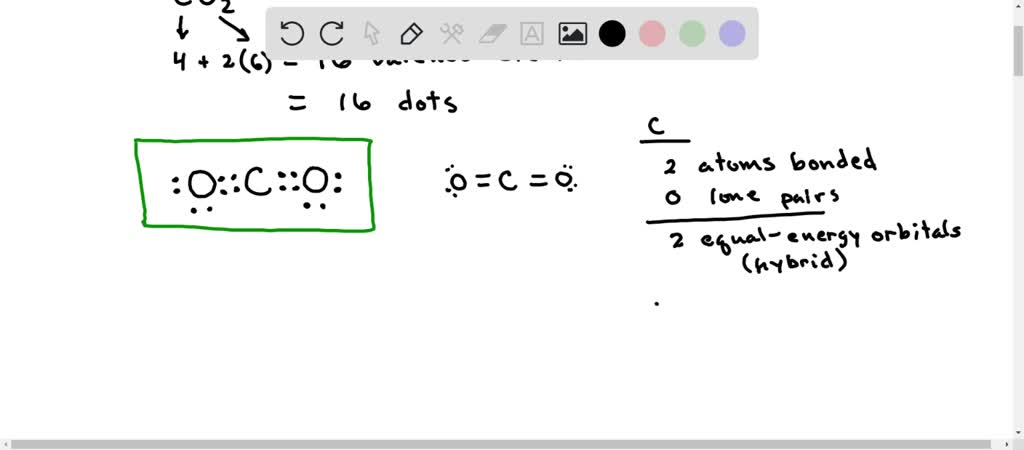
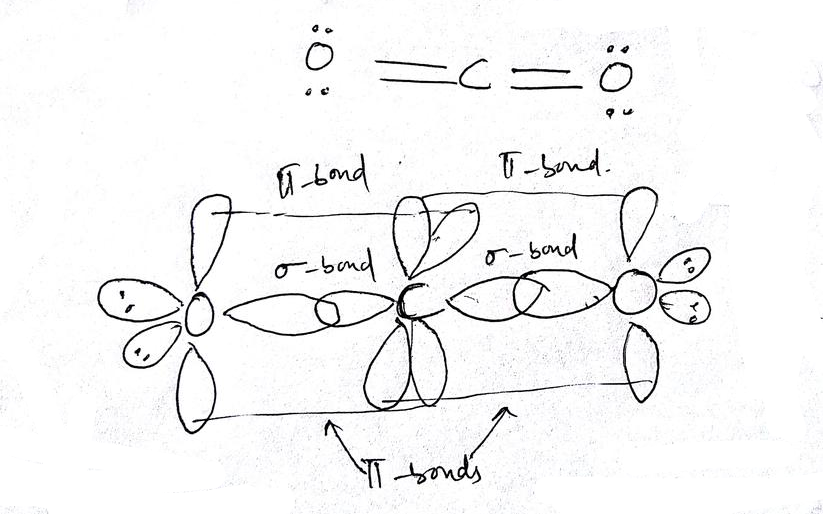
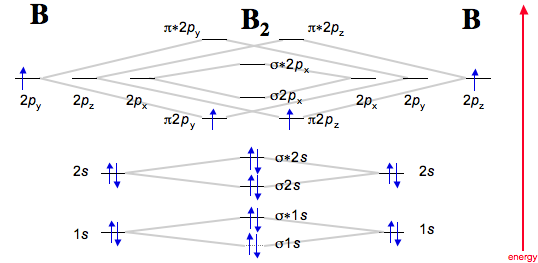
Molecular Orbital Diagram of Polyatomic CO2 Molecules - Chemical Bonding & Molecular Structures - YouTube

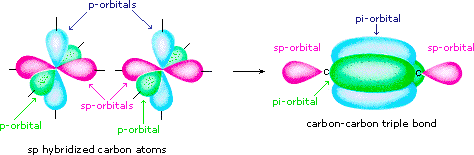
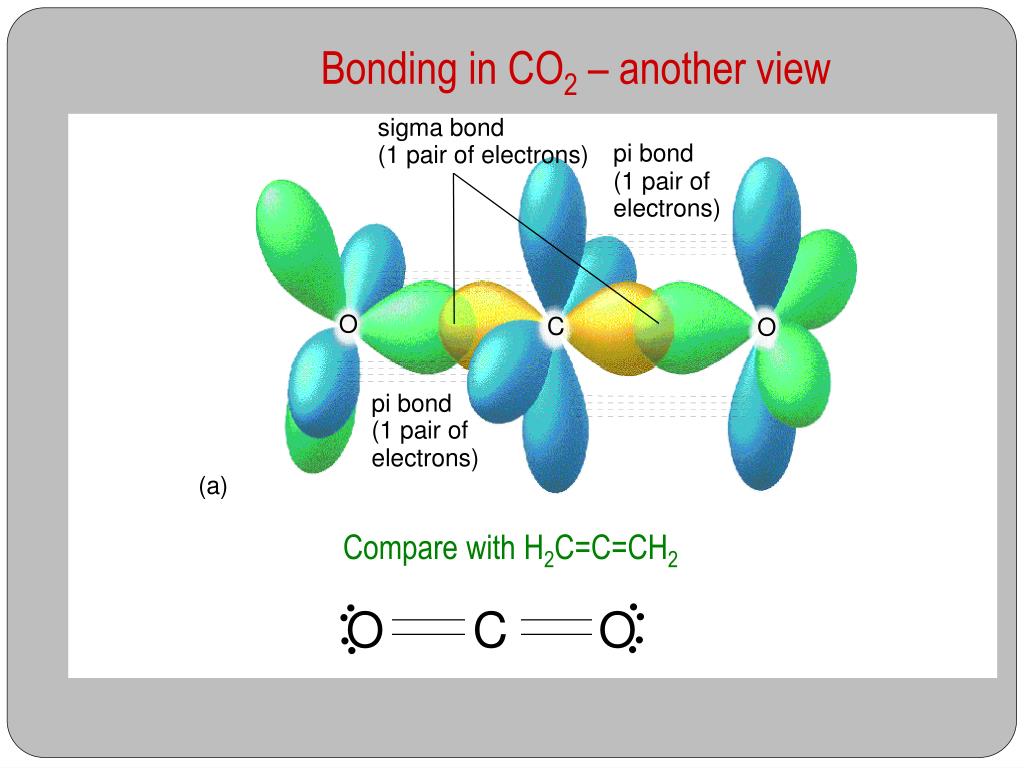
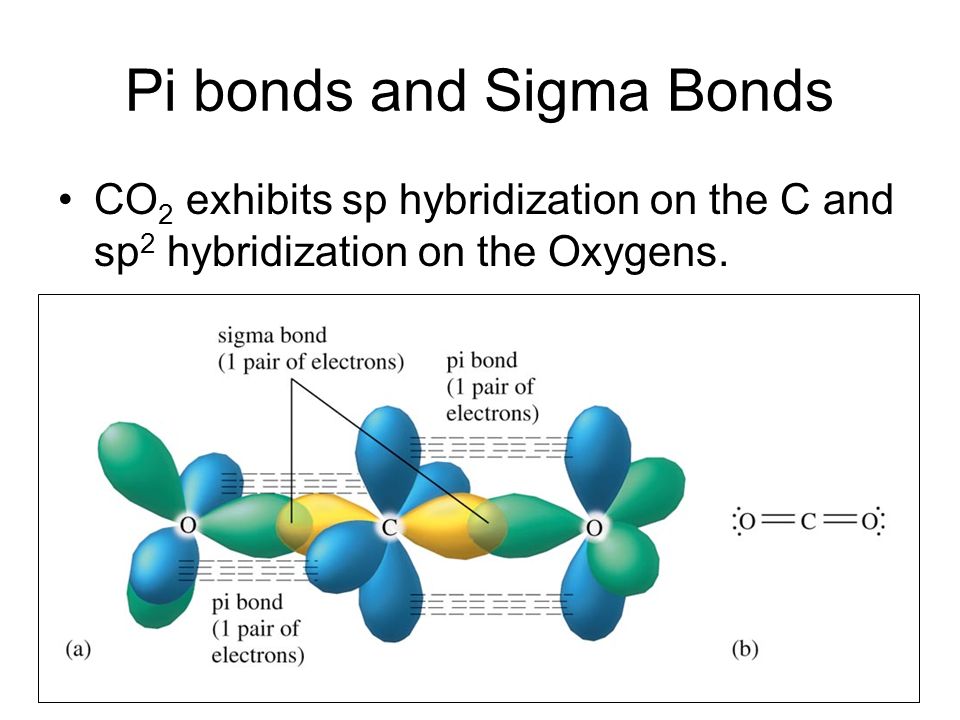
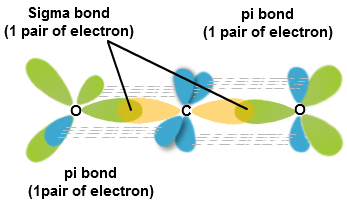
What atomic or hybrid orbitals make up the pi bond between C and O in carbon dioxide, CO_2 ? orbital on C + orbital on O How many o bonds does C

Dinitrogen and Carbon Dioxide Activation to Form C–N Bonds at Room Temperature: A New Mechanism Revealed by Experimental and Theoretical Studies | The Journal of Physical Chemistry Letters
1 Bonding Theories for Covalent Molecules (1): Valence Bond Theory VSEPR → predicts the 3-dimensional molecular (MG) and elec

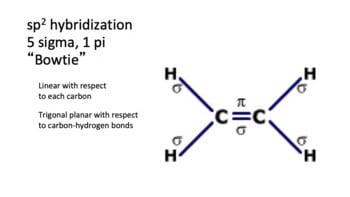
GLV Tutoring - In order to determine what type of hybridization is neccessary for each molecule follow these 3 easy steps: 1. Determine how many p orbitals will be needed (p-orbitals are